Is Clo2 Polar Lew Structure How To Draw The Lew Structure For Youtube
The molecule is linear, so the two bonds point in opposite directions. It is an important species in various. It is a highly reactive gas that consists of one chlorine atom and two oxygen atoms.
ClO2 Lewis Structure, Geometry, Hybridization, and Polarity
To determine whether the given molecules/ions are polar or. Clo2 is the molecular formula for chlorine dioxide. Chlorine dioxide (clo2) is a polar molecule.
Hclo2 is a polar molecule.
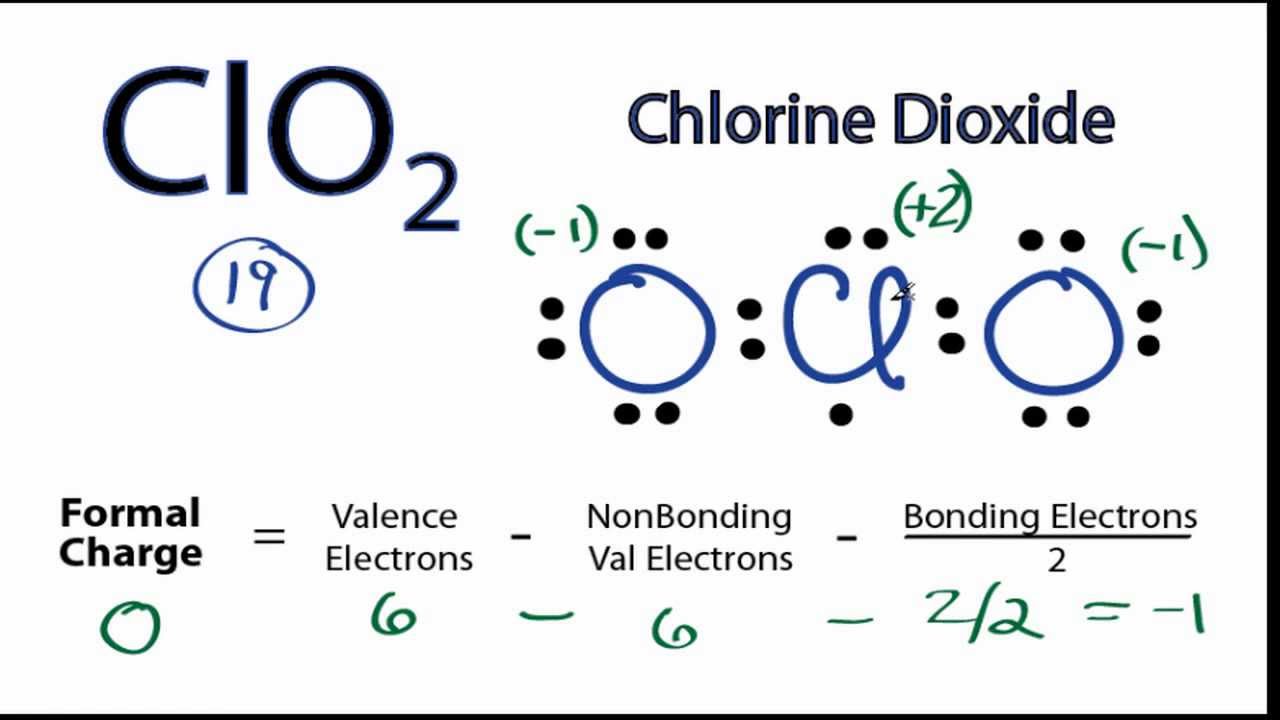
Calculate the total number of valence electrons for the molecule clo 2 −, including the extra electron because of the negative charge. The correct answer and explanation is : The bond polarity between two atoms can be estimated if. The oxygen atom with a single bond.
The structure of clo2 is represented by the molecular formula clo2. Clo2 is a potent oxidizing agent. It is polar, and oxygen is the atom closer to the negative side. The molecule has a bent shape and is.

This is because it contains polar bonds (due to differences in electronegativity between the atoms) and is not symmetrical in its molecular.
They are equally polar, so. Chlorite ion, denoted as clo₂⁻, is a polyatomic ion composed of a chlorine atom (cl) and an oxygen atom (o), carrying a negative charge.