Electron Geometry Of Nh4+ Lewis Structure How To Draw The Dot Structure For Ammonium
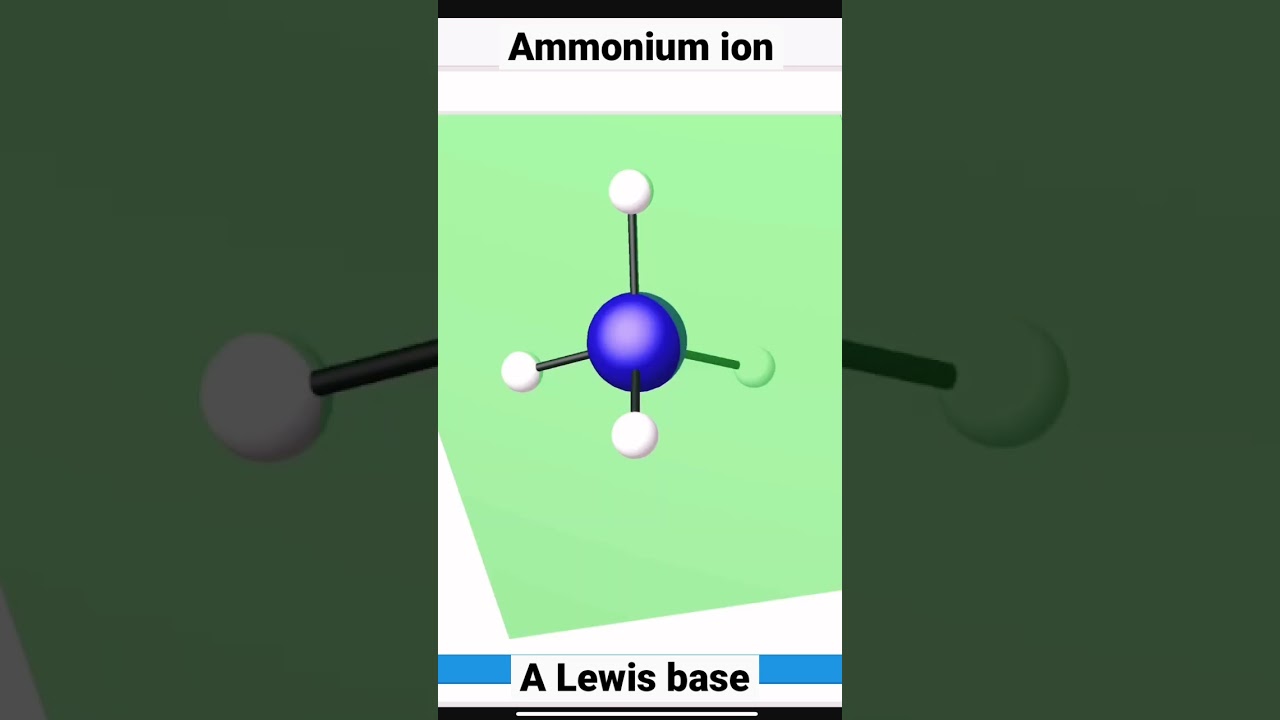
In the case of nh4+, the nitrogen atom does not have any lone pairs because all its valence electrons are involved in bonding with the hydrogen atoms. N has 5 valence electrons, and there are 4 h atoms, each contributing 1 electron. This means that the four electron pairs around the central nitrogen atom are arranged in a.
Nh4+ Lewis Structure Bond Angle Draw Easy
In the nh4+ ion, there are four hydrogen atoms bonded to a central nitrogen atom. We've got 4 of these, though; Let's do the lewis structure for nh4+, the ammonium ion.
The lewis structure of nh4+ can be represented as follows:
In this step, first calculate the total number of valence. The electron pair geometry for the ammonium ion (nh4+) is tetrahedral. By using the following steps, you can easily draw the lewis structure of nh 4+. There are 4 single bonds between the nitrogen.
First, we need to determine the number of electron pairs around the central atom (n) in nh4+. So nitrogen, on the periodic table, is in group 5, so it has 5 valence electrons. The bond angles in the molecule are approximately 109.5 degrees. Nh4+ (ammonium ion) lewis structure has a nitrogen atom (n) at the center which is surrounded by four hydrogen atoms (h).

Let’s one by one discuss each step in detail.
The electron geometry of nh4+ is tetrahedral, and the molecular shape is also tetrahedral.


+NH4%2B+Lewis+structure:.jpg)