Lewis Structure For Nh2+ Solved Draw The Nh2 Draw The Dot
The lewis structure for nh2, features one nitrogen atom bonded to two hydrogen atoms and carries a negative charge. This video shows you how to draw the nh2+ ion lewis dot structure. Both structures are correct as they are in resonance structure so you can choose any of them according to your ease or you.
Solved Draw the Lewis structure for NH2. Draw the Lewis dot
This arrangement suggests a trigonal planar geometry. The lewis structure of nh2 consists of a nitrogen atom in the center with two hydrogen atoms attached to it, and a positive charge on the nitrogen atom. This widget gets the lewis structure of chemical compounds.
Nh2 lewis structure# the most initial and important step towards finding out the bonding nature of any chemical molecular structure is to draw its lewis structure.
This is our ch(nh2)2+ final lewis structure diagram. Let’s one by one discuss each step in detail. Include all lone pairs or single electrons. Be sure to put brackets, along with a negative sign, around the nh.
The lewis structure of nh2+ (the amide ion) can be derived by considering the number of valence electrons and the need for the nitrogen atom to achieve a stable structure. Draw the lewis structure for nh2 draw the lewis dot structure by placing atoms on the grid and connecting them with bonds. It also discusses the bond angle, hybridization, and molecular geometry of the nh2+ ion. In this step, first calculate the total number of valence.
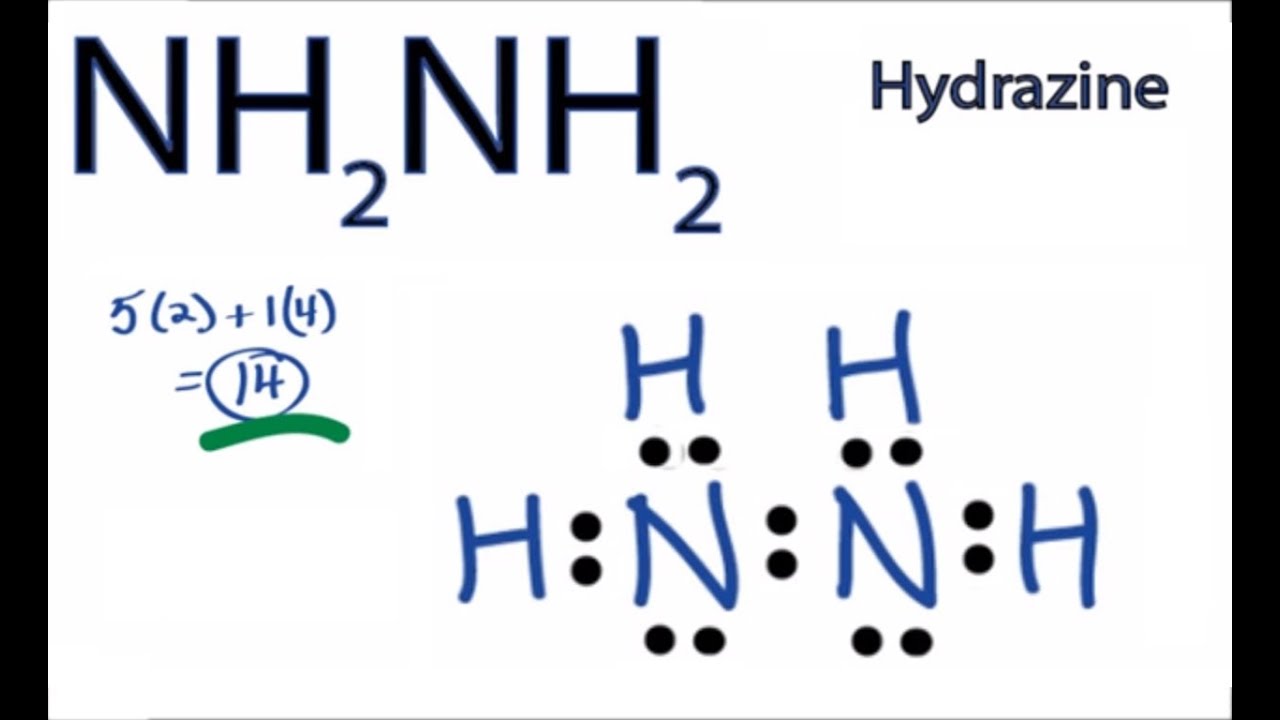
Get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle.
To draw the lewis structure and the.


