Here Are Sketches Of Four Electron Orbitals Solved D Yes Any
In your question about electron orbitals, you might be asking to identify the s orbitals and p orbitals from a group of sketches labeled a, b, c, and d. Your solution’s ready to go! Orbitals are not orbits, in the sense of being paths around the nucleus;
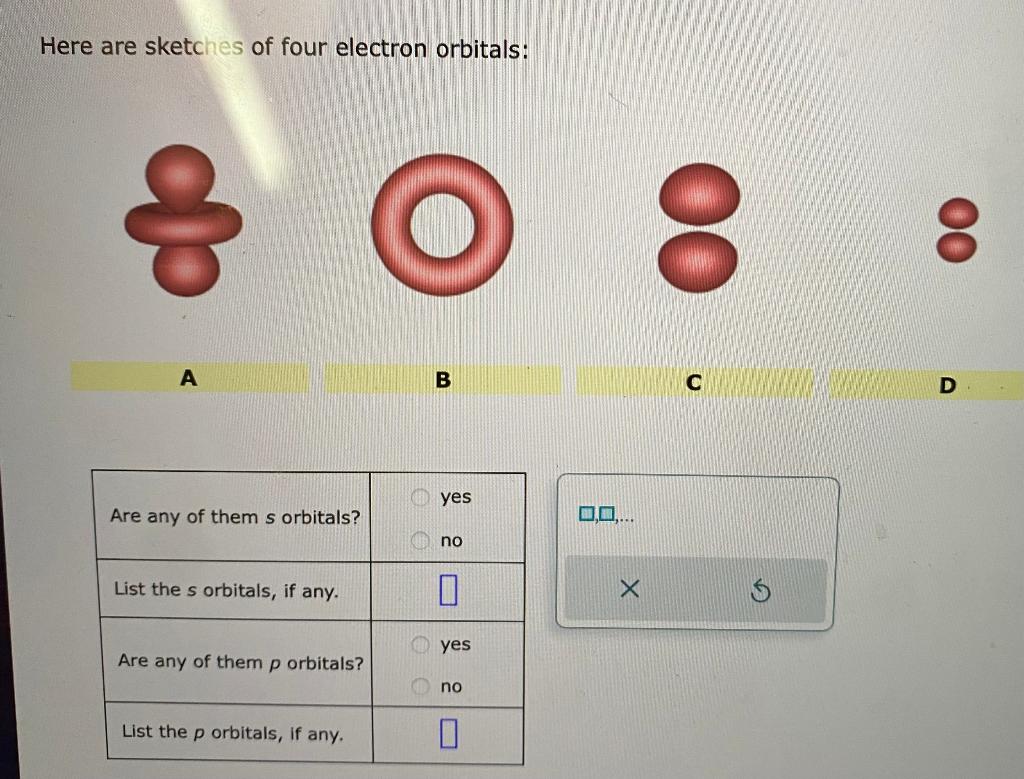
Solved Here are sketches of four electron orbitals 00 A B C
For each value of n,. We have to find s and p orbitals from sketches of four electron orbitals q: Use this information to answer the question below.
Whereas for p orbital, the shape is dumbbell like, which can be like this in y axis, flat on x axis and diagonal on z axis.
Among the given sketches, orbital a and orbital d are s orbitals, while orbital b and orbital c are p orbitals. There are 3 steps to solve this one. A penny has a mass of 2.50g and the moon has a mass of 7.35g *10^22kg. Solution for here are sketches of four electron orbitals:
For s orbital, the shape is spherical. To identify if any of the given electron orbitals are s. Understand the characteristics of s and p orbitals. Yes list the s orbitals, if any.

Be sure your answers have the correct number of significant digits.
Are any of them p orbitals? Are any of them p orbitals? List the p orbitals, if any. Instead, they represent the positions where the electron is most likely to be found.
Atomic orbitals are mathematical functions that describe the wave nature of electrons (or electron p. The findings were achieved using two methods: A are any of them s orbitals? Not the question you’re looking for?
Which of the following sets of quantum numbers can describe an electron in a 4s orbital?
Guy bartal's lab by dr. Here are sketches of four electron orbitals: A b c d are any of them s orbitals? List the s orbitals, if any.
In the quotient, we can.

