Is Glucose Nonpolar Solved Sugar A Polar Or Substance?
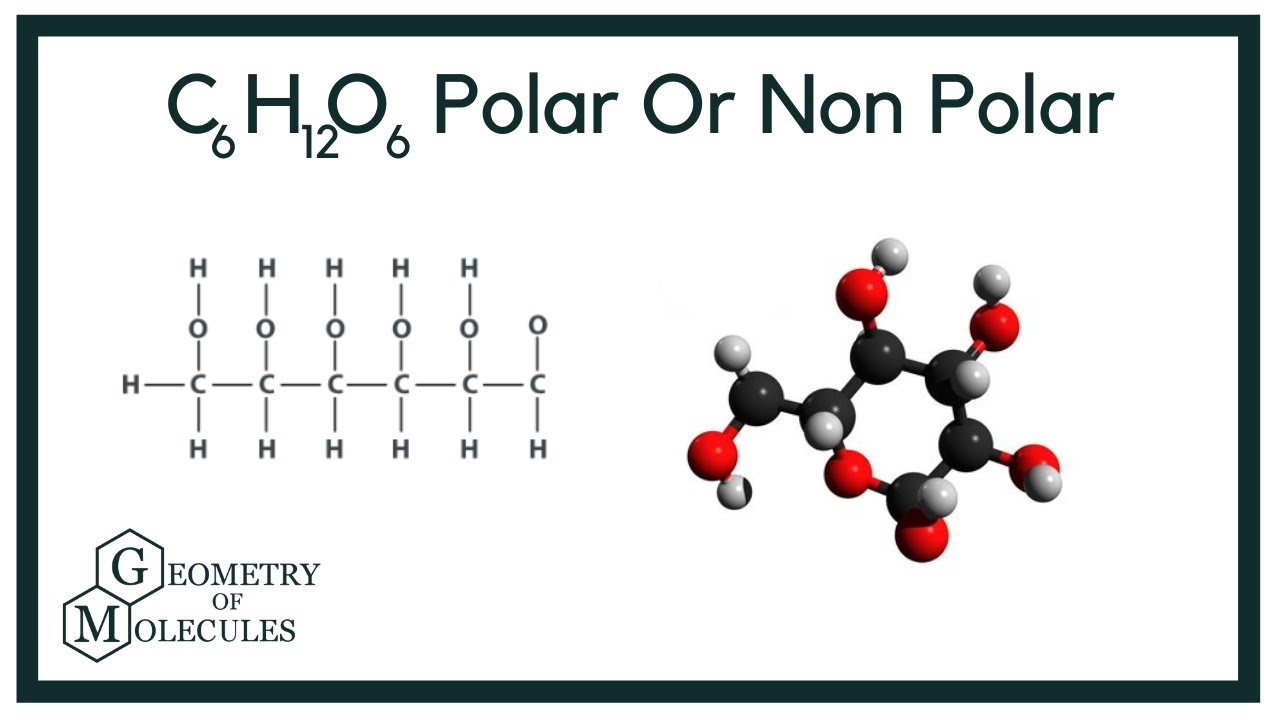
Sugars (e.g., glucose) and salts are polar molecules, and they dissolve in water, because the positive and negative parts of the two types of. The molecule consists of six carbon atoms, twelve hydrogen atoms, and six oxygen atoms. Glucose, $$ c_6h_{12}o_6$$, has $$ 4 $$ secondary hydroxyl groups, and $$ 1 $$ (exocyclic) primary hydroxyl group.
Is Glucose Polar or Nonpolar (C6H12O6) YouTube
Sugar is a polar covalent compound. The question of whether sugar is polar or nonpolar can be quite intriguing. Glucose, c_6h_12o_6, has 4 secondary hydroxyl groups, and 1 (exocyclic) primary hydroxyl group.
Glucose, c_6h_12o_6, contains 5 hydroxyl groups, and an ethereal ring junction.
Is saliva polar or nonpolar? By the end, you’ll understand why sugar is a special molecule, and why it’s neither polar nor nonpolar. Sugar is a highly polar molecule that has substantial water solubility. To address this directly, **sugar is polar**.
Sugar is a highly polar molecule that has substantial water solubility. Yes, glucose is a polar molecule. Sugar is a nonpolar molecule. Is a sugar molecule polar or nonpolar?
And thus this material is highly polar, and thus reasonably dissolves in the polar solvent.
Studies indicate that more than half of parotid and submandibular lipid secretions are nonpolar, while. The oxygen atoms in glucose have a higher. Sugar molecules do not have a permanent. Glucose is a polar covalent molecule because c, h, and o are all nonmetals, hence it being being polar since polar covalent.
This means that sugar molecules have an uneven. Co2 is non polar while so2 is polar molecule. Glucose is a polar compound. Is sugar polar or nonpolar?



