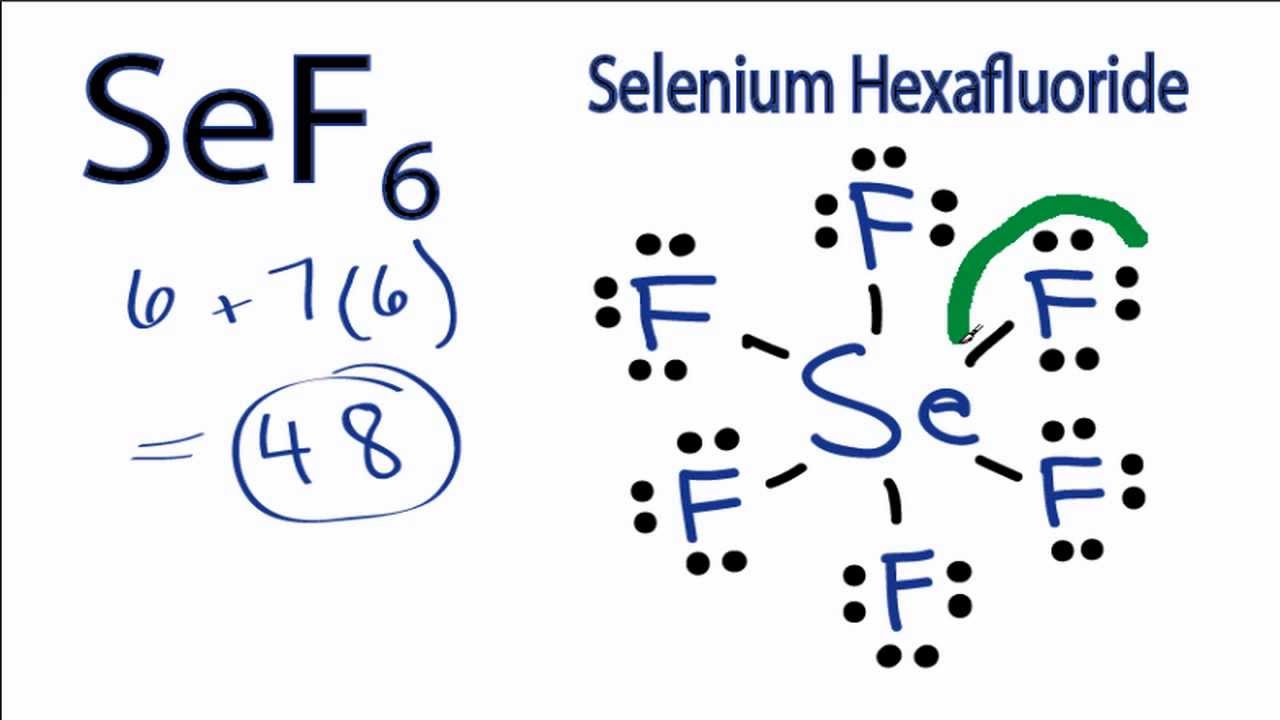
Hexafluoride Formula Sef6 Lewis Structure How To Draw The Lewis Structure For Selenium
The most common synthesis involves the direct reaction of sulfur with fluorine. This structure is also available as a 2d mol file; Sf 6 has an octahedral geometry,.
SeF6 Lewis Structure How to Draw the Lewis Structure for Selenium
Sulfur hexafluoride (sf 6) is a gas at standard temperature and pressure (25 °c, 1 atm). The structure of sulfur hexafluoride is octahedron, with 6 fluorine atoms. Before using the cylinder, ensure that the valve is closed, then remove the.
It is a hypervalent octahedral molecule.
The formula for xenon hexafluoride is xef6, where xe represents the xenon atom and the subscript 6 indicates the number of fluorine atoms bonded to it. Sulfur hexafluoride is an inorganic compound with the formula sf 6. Sulfur hexafluoride or sf6 is an inorganic, greenhouse gas. Several classes of compounds containing fluorine—including.
Sulfur hexafluoride (chemical formula sf 6) is a compound of sulfur and fluorine and the most potent greenhouse gas known. Supplied in a sure/pac ™ cylinder and has a brass needle valve with a male 1/4 nptf outlet thread installed.